Flucytosine Capsules, USP are indicated only in the treatment of serious infections caused by susceptible strains of Candida and/or Cryptococcus.
-
Compare toAncobon®*
Capsule fill: Corn starch, lactose monohydrate and talc.
The imprinting ink for both the 250 mg and 500 mg capsules contain black iron oxide.
The 250 mg capsule shell contains FD&C Yellow No.6, FD&C Green No. 3, D&C Yellow No. 10, titanium dioxide, sodium lauryl sulfate and gelatin.
The 500 mg capsule shell contains iron oxide yellow, iron oxide black, titanium dioxide, carboxymethylcellulose, sodium lauryl sulfate and gelatin.

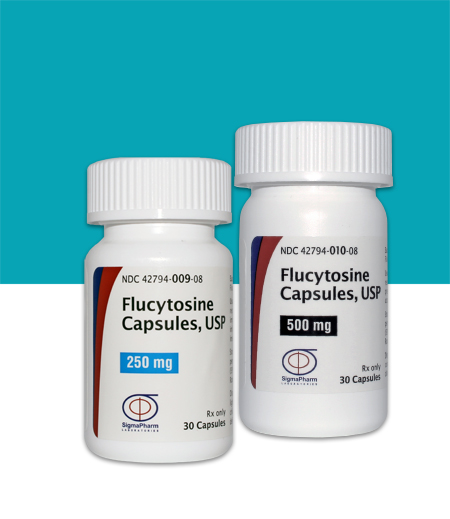
Flucytosine Capsules, USP (250 mg)
-
Package size30ct
-
Strength250 mg
-
NDC Code42794-0009-08

Flucytosine Capsules, USP (500 mg)
-
Package size30 ct
-
Strength500 mg
-
NDC Code42794-0010-08
