Our product selection and R&D efforts are almost exclusively focused on the identification and advancement of unique, difficult-to-formulate Generic and Branded products, which cannot be likely developed by most of our competitors. Our products are currently developed and manufactured using our own proprietary drug delivery systems, which can effectively optimize drug solubility, bioavailability and stability, and further, which can bypass, and avoid infringement of, Orange Book patents related to their corresponding reference listed drug products.
"Through our proprietary technology platforms, we identify and develop unique Generic and
Branded products that cannot often be developed or matched by our competitors"
Presently, our company owns or controls more than one hundred and twenty (120) patents and patent-publications worldwide. Several newer proprietary compositions and innovative formulation techniques, which are fully scalable and manufacturable, are also available only to us while being thoroughly protected as our trade secrets.
Sigmapharm’s “StereoSol-Amorphous,” “Micro-Mono-Disperse” and “Particulate-UniBlend” technologies, are some of our newer proprietary drug delivery systems used to successfully develop and manufacture Generic and Branded drug products that:
- Bypass, and avoid infringement of, crystalline-polymorph and particle-size patent claims;
- Maximize drug-powder blend uniformity and drug content uniformity;
- Possess optimized drug solubility, bioavailability and stability properties;
- Contain lower drug doses as compared to their reference products;
- Enhance drug efficacy and improve therapeutic dose regiments;
- Minimize or eliminate any Food Effects, and improve GI safety;
- Provide new or extended patent protection for either already approved and commercialized drugs or new chemical entities at their drug development stages; and/or
- Utilize the 505(b)(2) New Drug Application (Paper-NDA) regulatory path that often results in a faster and lower-cost FDA approval.
"Backed by our proprietary technologies, our personnel's proven innovative track record
and our state-of-the-art R&D, analytical chemistry and manufacturing facilities,
our company's potential for success is virtually unlimited"
We are proud to have one of the most innovative and efficient group of scientists and inventors in the industry, who have pioneered the development of several unique pharmaceutical products marketed domestically and internationally.
We are also proud to work and innovate, under highly effective and transparent quality systems, in our modern research and development, analytical chemistry and manufacturing facilities, which are fully equipped with state-of-the-art equipment and thoroughly updated and protected networks and computer-server systems.
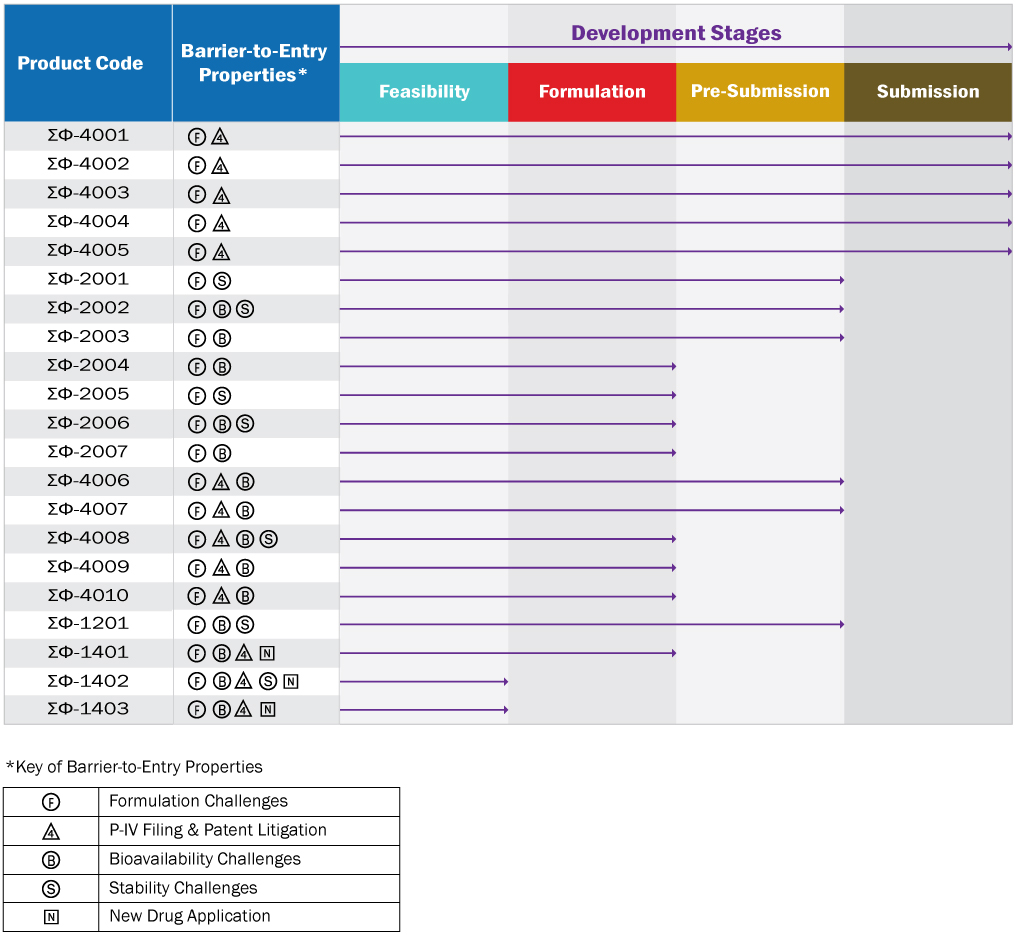
Pipeline of Unique Generic & Branded Products
Currently Prepared For ANDA & NDA Submissions
(Updated on October 5th, 2018)